Delegated IRB review is an exception to the general requirement of a review by a full panel of the IRB. Some research applications may be delegated to a single member of the IRB or a subgroup of the IRB when specific expertise is required. The IRB delegated review process is also referred to as the expedited review process. […]
Author Archives: Martin Letendre
Over the past six weeks, Veritas IRB has been assisting researchers and sponsors navigate through the challenges that the COVID-19 pandemic presents to their research. Over the coming days, we will share different issues that were presented to the IRB. This blog posting will address issues pertaining to obtaining and documenting free and informed consent. […]
In this period of national emergency, we remain committed to safeguarding the health and safety of our employees, customers, research partners, as well as the research participants under our supervision. Continuation of Services We plan to continue to offer full board IRB meetings on a weekly basis and delegated reviews of submissions on a daily […]
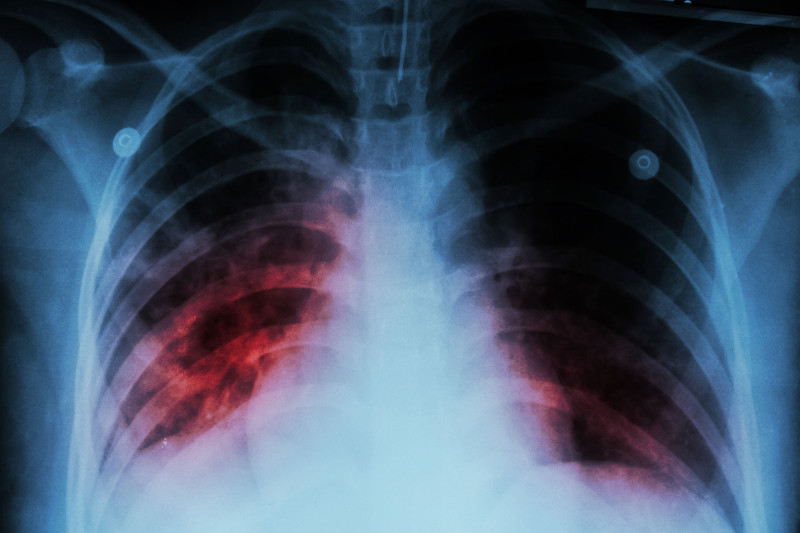
Following our March 3, 2016 post entitled “Preventing your study from becoming the next tragedy.”, we received numerous requests for information regarding the Montreal tuberculosis outbreak. This avoidable tragedy was not covered – to any great extent – by English Canadian media, which may explain why few Canadians have ever heard of it. It wasn’t […]
Every January, Las Vegas is home to the highly anticipated yearly conference called the Consumer Electronics Show. It’s difficult to believe that the CES is nearly 50 years old, as things have most certainly changed since 1967 on the technology front, and the conference has certainly been a substantial platform for many interesting consumers products, […]
Guest Blogger Andrew Milroy – HRPP and CQA Manager (Merita CQA) For 19 years ICH GCP E6 has not changed. Now, nearly two decades after its initial release, change is coming. Perhaps the biggest difference is the focus and preeminence of Quality Management. A modernized quality standard for clinical study processes is being espoused which […]